On April 2, the Centers for Medicare & Medicaid Services (CMS) released the 2019 Rate Announcement and Final Call Letter (RACL). The RACL is an annual regulatory policy document that establishes payment and coverage policies for Medicare Advantage (MA) and Part D plans for the upcoming plan year. The RACL incorporates proposed changes from both Part I and Part II of the 2019 Advance Notice and Call Letter (ANCL). CMS announced an upward revision to the growth rate which will positively impact health plan revenue. In the RACL, CMS largely finalized many of the changes proposed in Part II of the ANCL. CMS announced it will not implement the Payment Condition Count changes to the MA Risk Adjustment Model for PY 2019. It will instead look to implement the counts, required by the Comprehensive Addiction and Recovery Act of 2016 (CARA), starting in PY 2020.
Medicare Advantage Program Highlights:
According to CMS, county benchmarks will increase by 5.28% (up from 4.35% in the ANCL); the net change in plan revenues will be an increase of 3.40% (up from 1.84% in the ANCL) (see table below). In addition, for PY 2019, CMS expects that the MA coding trend will increase risk scores, on average, by 3.1%. Specifically, in the RACL, CMS:
- Makes minor changes to normalization factors: For PY 2019 CMS adopted a revised Part C normalization factor of 1.041 for the 2017 model and 1.038 for the CMS-HCC model (without condition counts). The lone factor change was to the RxHCC factor which was revised down slightly to 1.019.
- Declines to adopt condition counts in revised CMS-HCC risk adjustment model for CY 2019: While CMS adopted the inclusion of additional conditions for Chronic Kidney Disease, mental health, and substance use disorder in the CMS- HCC model, CMS chose to delay the proposed addition of condition counts. CMS interpreted CARA to allow the agency to delay the addition of condition counts until PY 2020 as even with the delay, CMS will still be able to phase-in the addition of condition counts over 3 years and have a final revised model by 2022 as required by CARA.
- Adopts an increased blend of Encounter Data System (EDS) In Risk Scores: For PY 2019, CMS will blend 75% of the risk score calculated with the 2017 CMS-HCC model and diagnoses submitted on RAPS records and FFS claims with 25% of the risk score calculated with the updated CMS-HCC model without count variables and diagnoses submitted on encounter data records, RAPS inpatient records, and FFS claims.
- Declines to adopt the updated RxHCC model: CMS will not implement the proposed update to the RxHCC Risk Adjustment Model in 2019 and will use the same model as used for 2018. The RxHCC model is used to adjust direct subsidy payments for Part D benefits offered by stand-alone Prescription Drug Plans (PDPs) and Medicare Advantage-Prescription Drug Plans (MA-PDs).
- Continues the categorical adjustment index (CIA) for duals: CMS will continue to apply a CAI to plan Star Ratings to address the impact of dual eligible and disabled members on plan performance. The 2019 CAI will include updated values and will include the additional “reducing the risk of falling” Part C measure. In 2019, the CAI will apply to 7 Part C measures and 2 Part D measures, 3 more than in 2018.
- Adopted a revised employer group waiver plan (EGWP) payment model: For 2019, CMS will 100% phase in the use of individual market plan bids to calculate the bid-to-benchmark ratios to set EGWP payments. CMS will adjust the individual market bids to account for the difference in the proportion of beneficiaries enrolled in Health Maintenance Organizations (HMOs) and Preferred Provider Organizations (PPOs) between EGWPs and individual market plans. However, CMS will seek comment on additional modifications for 2020 that include new or different adjustments for regional vs rural/local PPOs.
- Will largely retire the Beneficiary Access and Plan Performance Problems (BAPP) measure: CMS finalized its proposal to retire the current BAPP measure. CMS will include a revised BAPP, which only includes Compliance Activity Module (CAM) data, on the display page.
- Finalizes its scaled reductions for appeals IRE data completeness issues proposal: CMS has finalized its proposal to reduce a contract's Star Rating for data that are not complete or lack integrity using TMP or audit data. CMS will reduce star ratings commensurate with the degree of the data issue. CMS will implement the scaled reductions starting with the 2019 Star Ratings.
- Recognizes the permanent authorization of Special Needs Plans (SNPs): The Bipartisan Budget Act of 2018 (BBA) permanently reauthorized SNPs. CMS may make additional changes to SNPs as they review the BBA.
- Finalized the enrollment-weighting of Star Ratings in contract consolidations: CMS finalized the use of enrollment-weighting of the measure scores of the consumed and surviving contracts to calculate the ratings for the first and second plan years following the contract consolidations. The consolidation provisions will be applicable beginning with the 2020 QBPs and 2020 Star Ratings produced in fall 2019. CMS notes that this policy will not apply to situations where one entity is buying all or part of the business of another entity.
- Adopts new and updated Star Ratings: CMS adopts several new and changed Star Ratings for 2019:
- New Measures for 2019:
- Statin Use in Persons with Diabetes (SUPD)
- Statin Therapy for Patients with Cardiovascular Disease.
- Changes to Measures for 2019:
- CMS largely adopted the proposed updated list of measures to support the calculation of the Quality Improvement measure-- the lone change was the addition of “Reducing the Risk of Falling” measure
- CMS proposes a number of changes to non-clinical measures including several medication adherence measures, and the MPF Price Accuracy, Members Choosing to Leave the Plan, and the Reducing the Risk of Falling measures
- Changes to Existing Measures and Potential Measures for 2020 and Beyond:
- Updates to the Controlling High Blood Pressure measure
- Revisions to the HEDIS Plan All-Cause Readmissions measure
- Inclusion of telehealth and remote access visits in applicable measure calculations
- The potential to exclude individuals with advanced illness from select HEDIS measures
- Assessments of the quality of care transitions from an inpatient setting to home
- A measure to assess follow up care provided after an emergency department visit for patients with multiple chronic conditions
- An increased emphasis on care coordination measures, including a desire to develop new care coordination measures
- An assessment of members of the ability to control opioid overuse
- An expanded Care for Older Adults measure with more indicators and a broader population
- New Measures for 2019:
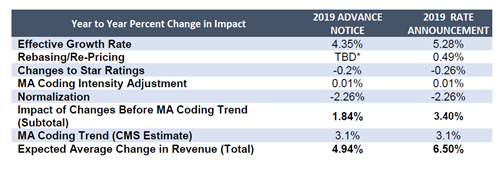
Highlights of the estimate above include:
- Growth rate: CMS estimates that FFS United States Per Capita Costs will increase by 5.11% from 2018 to 2019, to $891.07. CMS adjusts for county-level relative costs when calculating the benchmarks. Some county benchmarks are also capped at the amount that would have been paid pre-ACA.
- Star Ratings: For the second year in a row, CMS estimates that changes to plan Star Ratings will result in a decrease in payments to plans in 2019.
Part D Program Highlights:
- Part D benefit parameters increase by 1.94%: CMS will revise most of the statutory parameters for the defined standard Part D benefit, including the annual deductible, initial coverage limit, and annual out-of-pocket limit, by 1.94% for 2019.
- Specialty tier threshold will not increase: CMS will maintain the $670 specialty tier threshold in 2019. CMS notes that an analysis of CY 2017 prescription drug event (PDE) data showed that only 1% of 30 day-equivalent fills exceeded $670. However, CMS will monitor trends in use of drugs with costs exceeding the specialty tier threshold.
- Non-preferred brand tier composition: CMS adopts a maximum threshold of 25% generic composition for the non-preferred brand tier for CY 2019, based on agency concerns that including a significant proportion of generic drugs on a brand-labeled tier may lead to beneficiary confusion.
- Non-preferred drug tier outlier tests: CMS will continue to conduct outlier tests for plans using the non-preferred drug tier who choose a copay structure for that tier. Plans flagged as outliers must provide a justification that the cost-sharing structure provides value for beneficiaries. In addition, those plans must provide detailed information about the generic drugs on the non-preferred drug tier which may include expected utilization, formulary alternatives on lower tiers, and tier placement strategy for generic drugs. If the justification is not accepted, plans may have to modify their benefit structure or formulary tiering.
- Benefit review: CMS will continue to evaluate expected cost-sharing amounts incurred by beneficiaries under coinsurance tiers in the Part D benefit and compare them to the impact of copayments. Sponsors submitting coinsurance values for its non-specialty tiers that are greater than the standard benefit of 25% will be compared to the established copay thresholds to determine whether the coinsurance values are discriminatory. Similarly, CMS will evaluate the drug composition of copay tiers to ensure the structure provides a meaningful benefit.
- Medication Therapy Management (MTM): The 2019 MTM program annual cost threshold will be $4,044. The new threshold was set by increasing by 1.94%, the annual percentage increase for 2019, the 2018 threshold of $3,967.
- Access to Part D vaccines: Consistent with statements in prior Call Letters, CMS acknowledges low vaccination rates for Part D vaccines, and encourages plan sponsors to either offer a $0 vaccine tier or to place vaccines on a formulary tier with low cost-sharing.
- Access to Over-the-Counter (OTC) medications in Part D: CMS will not grant Part D plan sponsors additional flexibility to offer access to OTCs under basic prescription drug benefits or as a supplemental benefit under enhanced alternative coverage.
- Change in the Coverage Gap Discount Program: The BBA increased the manufacturer discount for beneficiaries in the gap from 50% to 70% and reduced beneficiary cost sharing to 25% in 2019. Although, those discounts will continue to count toward a beneficiary’s true out-of-pocket cost (TrOOP). In addition, biosimilars will no longer be exempt from the rebate program. However, CMS is concerned that these changes may lead to increased drug costs. CMS indicates that it will review changes in generic drug uptake, formulary inclusion, tier composition, and substitutions to see if changes are necessary to protect beneficiary out-of-pocket costs and federal spending.
- Modification of opioid management in Part D: CMS adopted a variety of new strategies to manage opioid overuse among beneficiaries, including:
- Requiring all Part D sponsors to implement a hard safety edit that limits initial opioid prescription fills used to treat acute pain to no more than a 7-day supply
- Enhancing the Overutilization Management System (OMS) by adding additional flags for high-risk beneficiaries using potentiator drugs (e.g., gabapentin) in combination with prescription opioids
- Implementing the drug management program authorized by CARA to authorize plan sponsors to “lock-in” at-risk beneficiaries to specific prescribers and pharmacies and to establish beneficiary-specific claims edits to limit access to certain medications
- Requiring Part D plan sponsors to implement real-time safety edits at the point of service (POS) to engage patients and prescribers on overdose prevention
- Requiring all Part D plan sponsors to implement an opioid care coordination edit at 90 MME per day (instead of the proposed hard edit at 90 MME in the ANCL)
- Expecting sponsors to implement soft POS safety edits based on duplicative therapy of multiple long-acting opioids
- Implementing technical revisions to the Pharmacy Quality Alliance (PQA) opioid overuse measures
Access to Medication-Assisted Treatment (MAT): CMS will continue to examine sponsor formulary inclusion, utilization management criteria, and cost-sharing of Part D drugs indicated for MAT. CMS expects Part D sponsors to include products in preferred formulary tiers and to not place generic MAT drugs in brand tiers. CMS will not approve prior authorization (PA) criteria requiring a beneficiary to need an authorization for buprenorphine MAT more than once a year.
Avalere's Take
For Part D, CMS’ largely finalized the minor changes proposed in the ANCL as compared to previous plan years. However, CMS finalized significant changes to both Part C payment and operations as compared to its proposals in the ANCL. The Part C changes will increase projected payments to MA plans while holding back some of the model changes that could have raised operational concerns.
In looking at the MA program, CMS largely adopted many of the changes desired by MA plan sponsors. CMS finalized its new interpretation of uniformity requirements—giving plans greater flexibility in benefit provisions to enrollees with defined disease conditions. CMS also largely adopted as proposed its extension of supplemental benefits to non-medical maintenance items if they compensate for physical impairments, diminish the impact of injuries or health conditions, and/or reduce avoidable emergency room utilization. In addition, CMS chose not to add condition count changes to the risk adjustment formula for 2019—choosing instead to delay the addition to 2020 so that CMS can more comprehensively study the impacts of the change. Finally, CMS chose to largely retire the BAPP measure, and will apply a one-year exemption that will allow a CPE program audit to satisfy the sponsoring organization’s annual internal compliance program audit in the subsequent calendar year. However, CMS did move forward with the transition to using an increased blend of encounter data in the CMS-HCC model—raising the EDS blend from 85% RAPS / 15% EDS to 75% RAPS / 25% EDS. Plan sponsors remain skeptical about the accuracy and reliability of EDS data given data collection concerns.
